Mitox™ DNA - Mitochondrial toxicity assay
Helping improve quality of life
General information
Good physical condition is one of the most important parameters determining quality of life. In healthcare and in sports, being able to easily obtain a precise indication of the condition of patients and athletes enables physicians to customize treatment and care for optimum quality of life and performance. There are many ways to evaluate physical condition beyond relying on patients' anecdotal reports and physician observation. Physical capability and endurance tests, augmented by common blood tests provide a top line picture. However, to accurately determine current physical condition and foresee upcoming changes, physicians need to understand what is going on at the molecular level.
One of the key ways to obtain a precise and accurate picture of physical condition is to investigate mtDNA. Mitochondria are the organelles that provide energy to cells. Disturbances in the mitochondria undoubtedly result in changes to the energy-metabolism of the cell.
With the Retina Mitox™ assay, Primagen is the first company to develop accurate and reproducible assessment of mtDNA and mtRNA to the market. As a result of its proprietary position, the Company has been awarded by the United States Patent and Trademark Office in November of 2005, critical patents on its technology which will strengthen its commercial efforts in establishing the Mitox™ brand in the pharmaceutical drug discovery / research, clinical research and laboratory service markets in the field of mitochondrial measurement.
Antiviral therapy can cause serious side effects that impair the quality of life of patients receiving anti-viral drugs; NRTIs in particular can induce mitochondrial toxicity and contribute to several HIV, HCV, and oncology therapy-related complications. Mitochondria are unique in having their own replication enzyme, DNA polymerase gamma. The NRTIs affect the function of this enzyme, leading to the depletion of mtDNA. Such inhibition of the mtDNA synthesis is the mechanism of several known toxicities of nucleoside analogues: myopathy, cardiomyopathy, peripheral neuropathy, pancreatitis, lactic acidosis, hepatic steatosis and lipodystrophy (the “adverse effects”).
| Nucleoside Analogues | Common adverse effects |
| Zidovudine (Retrovir) GlaxoSmithKline (1987) | GI intolerance, headache, insomnia, asthenia. Bone marrow Suppression, anemia, neutropenia |
| Didanosine (Videx) Bristol-Myers Squibb (1991) | Nausea/Diarrhea, bloating.Pancreatitis, Peripheral neuropathy. Cautious use with stavudine in pregnant women |
| Zalcitabine (Hivid) Hoffmann-La Roche (1992) | Peripheral neuropathy; Stomatitis |
| Stavudine (Zerit) Bristol-Myers Squibb (1994) | Pancreatitis, Peripheral neuropathy. Cautious use with didanosine in pregnant women |
| Lamivudine (Epivir) GlaxoSmithKline (1995) | Minimal adverse effects/Toxicity |
| Abacavir (Ziagen) GlaxoSmithKline (1998) | Nausea, GI disturbances. Hypersensitivity reactions (Potentially fatal upon rechallenge) |
| Tenofovir Disoproxil fumarate (Viread) Gilead Sciences (2001) | Asthenia, headache, diarrhea, nausea, vomiting, flatulence, renal disorders |
With the increasing number of patients on long-term anti-viral therapy, drug-associated toxicity is one of the biggest problems in treating AIDS patients and is increasingly sobering the initial enthusiasm regarding increased therapeutic effectiveness in the treatment of AIDS.
The Mitox™ assay provides an effective tool for predicting and monitoring mitochondrial toxicities in infected patients receiving NRTIs or other replication inhibiting drugs, such as certain oncology drugs. The assay can assist MDs and patients in deciding to change therapy before adverse side effects develop. The assay can also be used in the early stage of drug development to detect mitochondrial toxicities of the compounds before they are made available on the market. Use of this assay during drug development and at regular intervals during treatment will contribute to a better quality of life for patients who are treated with these drugs.
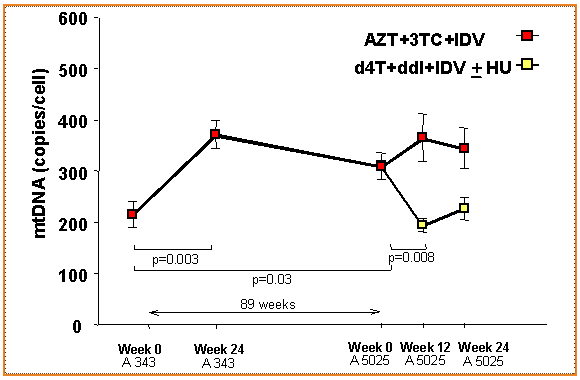
The Mitox™ test is clinically validated in a number of different studies. A typical example is shown in the figure below.
Tebas et al In press: The effects of antiretroviral therapy on mitochondrial DNA (copy number per cell) in peripheral blood mononuclear cells. After 89 weeks of treatment on AZT+3TC+IDV (n=14) subjects were randomized to continue their antiretroviral regimen (red) (n=5) or to switch to DDI+D4T+IDV + HU (white) (n=9). The values represent mean + SE. After initiation of therapy there is a significant increase in the amount of mitochondrial DNA, which decreases significantly after the switch of nucleosides.
Over time the toxic effects of drugs evolve at the mitochondrial level, specifically if patients switch to drugs that are known to be more toxic than others (bifurcation of the curve). The patients in the lower arm of the curve have the highest risk of experiencing toxic side effects.
In vitro and in vivo experiments with nucleoside analogue compounds have demonstrated that during the course of treatment with these drugs, there is a direct link of toxic adverse effects to the number of mitochondria per cell. Typically, a loss of 45%-60% of mitochondrial function is observed prior to these toxic events. While most patients on these drugs actually demonstrate an increase in mitochondrial function within 4-8 weeks of therapy, due in part to a compensatory reaction to pressure on the production of mitochondrial, generally, patients who will have higher likelihood of adverse effects will see quick loss of mitochondrial function in this same time period. As time goes by the required energy per cell cannot be supplied anymore via the normal mitochondrial pathways, and adverse effects will manifest.
Other Mitox™ applications
While early commercial and research collaborations have focused on assessment of human mitochondrial measurement, as a biomarker for toxicity and/or efficacy of antiviral treatment, Primagen has investigated several additional opportunities in the clinical field.
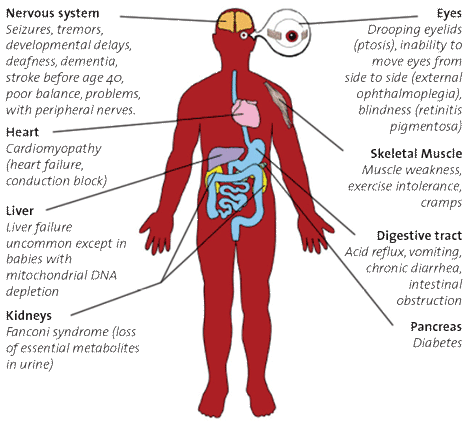
Mitochondrial disturbances impact organs throughout the body, producing a wide range of symptoms.
Active investigations are on-going in the field of sepsis, male (in)fertility, diabetes and chronic fatigue syndrome. In all these areas the Company has established collaborations with academic and clinical institutes to assess the potential of these opportunities.
Mitox quantification assay on PBMC (standard)
The crucial importance and utility for the Mitox™ test is its ability to predict future toxic effect and therefore enabling adjustment of therapy management prior to the onset. We also expect that prolonged use of antiviral compounds known to be more toxic versus newer generation compounds might extend the useful lifecycle of these drugs. This can extend the number of antiviral choices the patient has in managing their disease and it may serve well the economic consequences in managing antiviral drugs.
The Mitox test measurers mtDNA and nuclear DNA in a single assays allowing quantification of the number of mtDNA copies per cell through the ratio of both targets. The single tube, two marker, and real-time assay is i) robust by its design, ii) reduces the risk of contamination and iii) can easily be used in a high throughput systems.
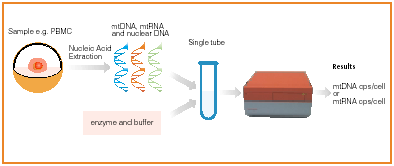
Retina Mitox duplex test procedure
The development of the Mitox™ mtDNA kit was completed in 2004 by receiving its CE mark. The Company currently uses this kit for its clinical research fee for service business based in Amsterdam, The Netherlands. We have commenced sales and distribution of test-kits to strategic partners in the USA and Europe. The Mitox™ mitochondrial RNA test is currently available as a lab service only.
Mitox quantification assay on human tissue (liver, fat, muscle, hart, etc.)
Much basic clinical research is done by verifying the target organs directly or via biopsies. If non-invasive diagnostic methods are lacking, physicians have no choice than performing diagnostics via biopsy. If biopsies have to be taken, one would like to test as many parameters as possible. At Primagen the Mitox assay can be fine tuned to any type of tissue. We have a proven track record on liver, adipose tissue, kidney and muscle biopsies.
Mitox quantification assay on cultured cells (in-vitro study)
In addtion to our Retina Mitox™ DNA and RNA Assay which measures the mtDNA or mtRNA content per cell in a real time duplex format, we also offer testing services for our in vitro mitochondrial toxicity program.
The Retina Mitox™ Assay can save valuable time and resources when screening for mitochondrial toxicities. With our in-vitro toxicity program we can predict potential mitochondrial toxicity outcomes at the earliest possible stage of drug development (phase I or II).
Each of our mitochondrial tests is fully validated, reproducible and offers high throughput for reliable results. When combined with our cell culture expertise, Primagen can meet your drug development, pre-clinical IND requirements and ultimately your clinical program needs.
Our Service Testing department will provide you a first class scientific style report and meet critical time lines to keep your program on schedule.
mtDNA quantification assay on Rat and/or Mouse cells or tissue
Leveraging, the company's reputation in the field of mitochondrial toxicity in scientific publications and promotions at conferences has led to interest of the pharmaceutical industry to use the Mitox™ test early in de the development of new drugs. Animal models are often used in early drug validation studies.
In a few cases where new drugs have made it to the clinical phase, Mitox™ is used in the follow up phase II and phase III clinical trials. Upon success of these trials the Mitox™ test may join in with the new drug as a combination for theranostics applications (i.e. close monitoring of treatment with dedicated diagnostics).
IP position
As a result of its proprietary position, the Company has been awarded by the United States Patent and Trademark Office in November of 2005, critical patents on its technology which will strengthen its commercial efforts in establishing the Mitox™ brand in the pharmaceutical drug discovery / research, clinical research and laboratory service markets in the field of mitochondrial measurement.
“A method of determining whether a medicament has therapeutic activity and/or possible side-effects, said method comprising: introducing a medicament to an organism; determining in a sample obtained from said organism a relative ratio of a mitochondrial nucleic acid and/or gene product thereof to a chromosomal nucleic acid and/or gene product thereof; and determining whether there is a change in the relative ratio during and/or after introduction of the medicament, wherein said change in said relative ratio is indicative that said medicament has therapeutic activity and/or possible side-effects”
Licenses
Patents and other IP rights are critical to Primagen's business. The Company's policy is to file patent applications to protect its product concepts and candidates, improvements thereto that may be important for the development of the Company's business. More specifically, Primagen will continue to aggressively file patent applications regarding its product candidates and concepts as well as their potential applications for diagnostic application as broad as possible. The Company aims to broadly cover new aspects of its product candidates and concepts by filing follow-up patent applications to its existing patent and patent applications, as well as by filing patent applications to potentially limit the freedom to operate of the Company's competitors. In general, the Company aims to protect any invention that holds future value for in-house development or for out-licensing. Primagen's policy is to seek protection of its inventions in North America, Europe and in selected other countries.
Printable version of the Retina™ Mitox™ Antiretroviral therapy brochure